C1: Demonstrate a knowledge of dehydration synthesis and hydrolysis as applied to organic monomers and polymers.
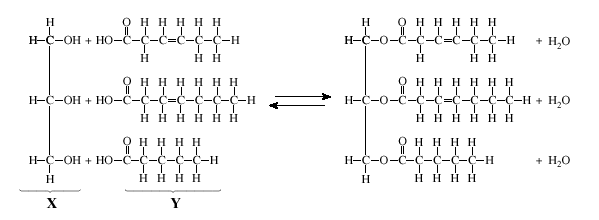
COMBINING: When making a polymer, water is removed from the reactants. This is known as dehydration synthesis. (Dehydrate means lose water. Synthesis means put together)
SPLITTING: When making monomers, water is added to the reactant. This is known as hydrolysis. (Hydro means water. Lysis means splitting)
C2: Differentiate among carbohydrates, lipids, proteins, and nucleic acids with respect to chemical structure.
Carbohydrates: Carbohydrates are one of the main types of nutrients. They are the most important source of energy for your body. Your digestive system changes carbohydrates into glucose (blood sugar). Your body uses this sugar for energy for your cells, tissues and organs.
Lipids: Lipids are a group of naturally occurring molecules that include fats, waxes, sterols, fat-soluble vitamins (such as vitamins A, D, E, and K), monoglycerides, diglycerides, triglycerides, phospholipids, and others.
Proteins: Proteins are large, complex molecules that play many critical roles in the body. They do most of the work in cells and are required for the structure, function, and regulation of the body's tissues and organs.
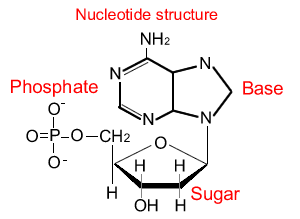
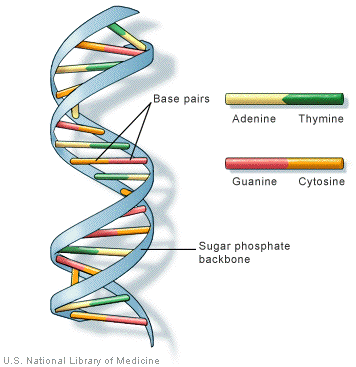
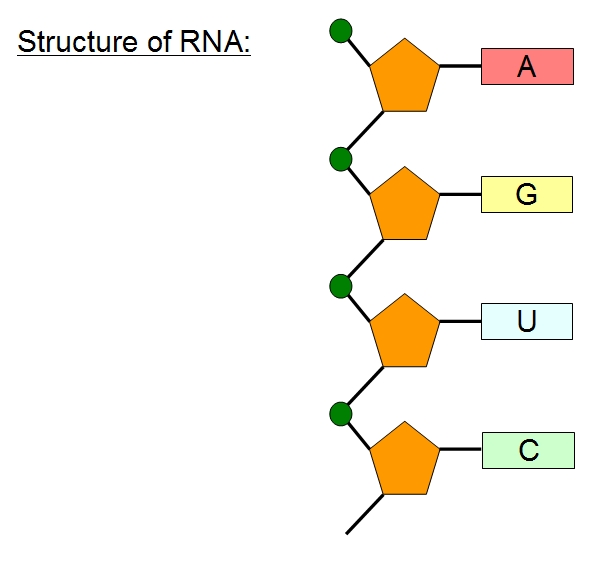
Nucleic Acids: Nucleic acids are biopolymers, or large biomolecules, essential for all known forms of life. Nucleic acids, which include DNA (deoxyribonucleic acid) and RNA (ribonucleicacid), are made from monomers known as nucleotides.
COMBINING: When making a polymer, water is removed from the reactants. This is known as dehydration synthesis. (Dehydrate means lose water. Synthesis means put together)
SPLITTING: When making monomers, water is added to the reactant. This is known as hydrolysis. (Hydro means water. Lysis means splitting)
C2: Differentiate among carbohydrates, lipids, proteins, and nucleic acids with respect to chemical structure.
Carbohydrates: Carbohydrates are one of the main types of nutrients. They are the most important source of energy for your body. Your digestive system changes carbohydrates into glucose (blood sugar). Your body uses this sugar for energy for your cells, tissues and organs.
Lipids: Lipids are a group of naturally occurring molecules that include fats, waxes, sterols, fat-soluble vitamins (such as vitamins A, D, E, and K), monoglycerides, diglycerides, triglycerides, phospholipids, and others.
Proteins: Proteins are large, complex molecules that play many critical roles in the body. They do most of the work in cells and are required for the structure, function, and regulation of the body's tissues and organs.
Nucleic Acids: Nucleic acids are biopolymers, or large biomolecules, essential for all known forms of life. Nucleic acids, which include DNA (deoxyribonucleic acid) and RNA (ribonucleicacid), are made from monomers known as nucleotides.
C3: Recognize the following molecules in structural diagrams.
ATP
ATP
DNA
Disaccharide
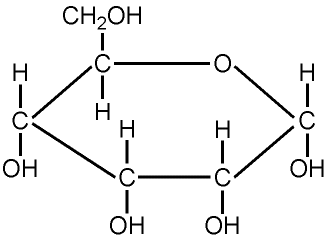
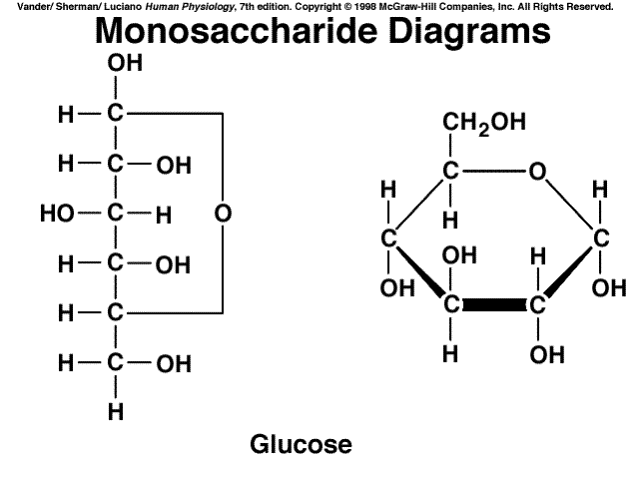
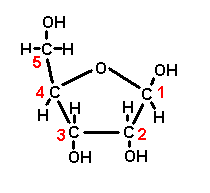
Glucose
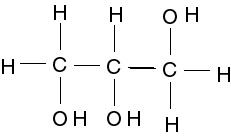
Glycerol
Hemoglobin
Monosaccharide
Neutral Fat
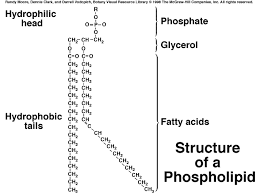
Phospholipid
Ribose
RNA
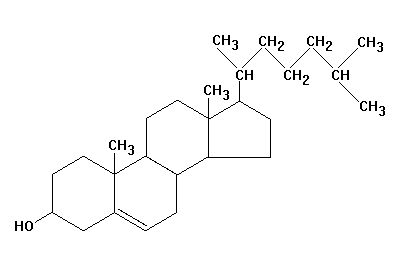
Steroids
C4: Recognize the empirical formula of a carbohydrate.
Carbohydrate molecules are characterized by the presence of the atomic grouping CH2O, in which the ratio of hydrogen atoms (H) to oxygen atoms (O) is approximately 2:1. All Carbohydrates have the general formula "CnH2nOn"
i.e. Glucose: C6H12O6
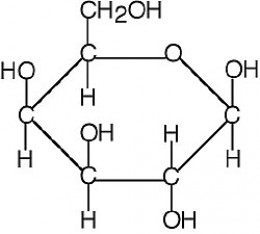
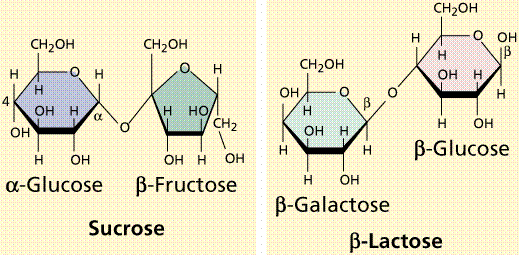
C5: Differentiate among monosaccharides, disaccharides, and polysaccharides.
A monosaccharide is the simplest sugar, a sugar building block. This is something like glucose, fructose, or galactose. It's a carbohydrate that cannot be decomposed by hydrolysis.
If you put two monosaccharides together and let them bond, they form a disaccharide, like maltose or sucrose or lactose.
A polysaccharide is basically a chain of monosaccharides bonded together. This is something like cellulose
C6: Differentiate among starch, cellulose, and glycogen.
Starch is a storage polysaccharide found in plants that is composed of glucose molecules. Cellulose is a polysachharide composed of glucose molecules and the chief constituent of a plant’s cell wall. Glycogen is a storage polysaccharide found in animals that is composed of glucose molecules joined straight but having numerous branches.
C7: List the main functions of carbohydrates.
The primary role of carbohydrates is to supply energy to all cells in the body. Many cells prefer glucose as a source of energy versus other compounds like fatty acids. Some cells, such as red blood cells, are only able to produce cellular energy from glucose.
C8: Compare ad contract saturated and unsaturated fats.
Saturated
1. solid at room temperature
2. found mostly in animals
3. no double bonds between carbons
Unsaturated
1. liquid at room temperature
2. found mostly in plants
3. double bonds found between carbons
Both saturated and unsaturated fats are composed of glycerol and 3 fatty acids.
C9: Describe the location and explain the importance of the following human body.
- Neutral Fats: are used for long-term storage, it insulates against heat loss, and it forms a protective cushion around major organs.
- Steroids: are lipids having a structure that differs entirely from that of fats. Steroid molecules have a backbone of four fused carbon rings, but each one differs by the arrangement of the atoms in the rings.
- Phospholipids: are not electrically neutral as are fats because the phosphate group can ionize. It forms the so-called "head" of the molecules while the rest of the molecule becomes the non-polar "tail."
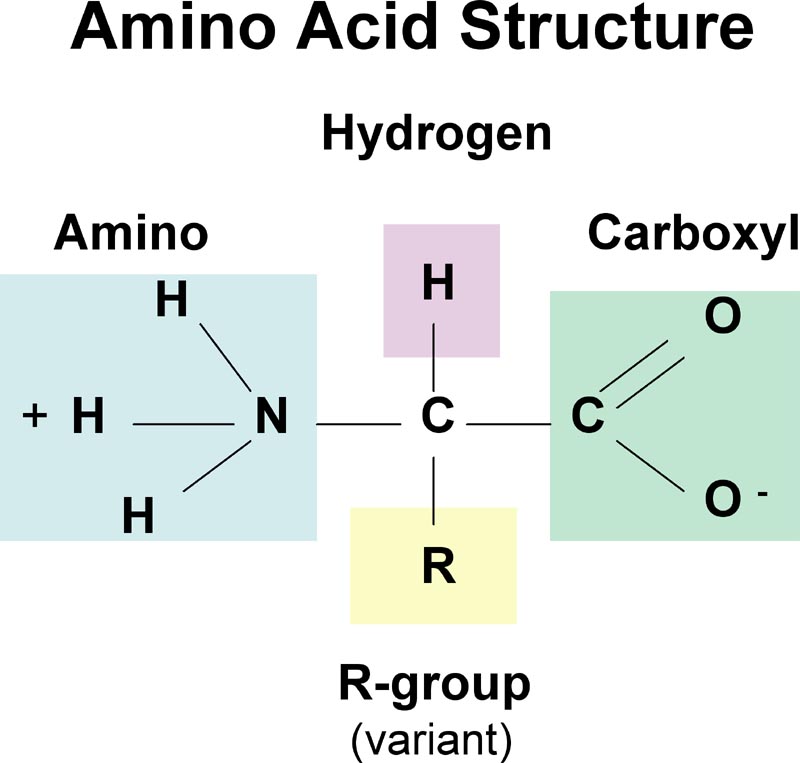
C10: Draw a generalized amino acid and identify the amine, acid and R groups.
Carbohydrate molecules are characterized by the presence of the atomic grouping CH2O, in which the ratio of hydrogen atoms (H) to oxygen atoms (O) is approximately 2:1. All Carbohydrates have the general formula "CnH2nOn"
i.e. Glucose: C6H12O6
C5: Differentiate among monosaccharides, disaccharides, and polysaccharides.
A monosaccharide is the simplest sugar, a sugar building block. This is something like glucose, fructose, or galactose. It's a carbohydrate that cannot be decomposed by hydrolysis.
If you put two monosaccharides together and let them bond, they form a disaccharide, like maltose or sucrose or lactose.
A polysaccharide is basically a chain of monosaccharides bonded together. This is something like cellulose
C6: Differentiate among starch, cellulose, and glycogen.
Starch is a storage polysaccharide found in plants that is composed of glucose molecules. Cellulose is a polysachharide composed of glucose molecules and the chief constituent of a plant’s cell wall. Glycogen is a storage polysaccharide found in animals that is composed of glucose molecules joined straight but having numerous branches.
C7: List the main functions of carbohydrates.
The primary role of carbohydrates is to supply energy to all cells in the body. Many cells prefer glucose as a source of energy versus other compounds like fatty acids. Some cells, such as red blood cells, are only able to produce cellular energy from glucose.
C8: Compare ad contract saturated and unsaturated fats.
Saturated
1. solid at room temperature
2. found mostly in animals
3. no double bonds between carbons
Unsaturated
1. liquid at room temperature
2. found mostly in plants
3. double bonds found between carbons
Both saturated and unsaturated fats are composed of glycerol and 3 fatty acids.
C9: Describe the location and explain the importance of the following human body.
- Neutral Fats: are used for long-term storage, it insulates against heat loss, and it forms a protective cushion around major organs.
- Steroids: are lipids having a structure that differs entirely from that of fats. Steroid molecules have a backbone of four fused carbon rings, but each one differs by the arrangement of the atoms in the rings.
- Phospholipids: are not electrically neutral as are fats because the phosphate group can ionize. It forms the so-called "head" of the molecules while the rest of the molecule becomes the non-polar "tail."
C10: Draw a generalized amino acid and identify the amine, acid and R groups.
C11: Identify the peptide bonds in dipeptides and polypeptides.
Peptides are short polymers formed from the linking, in a defined order, of α-amino acids. The link between one amino acid residue and the next is known as an amide bond or a peptide bond.
A dipeptide is a molecule consisting of two amino acids joined by a single peptide bond.
Proteins are polypeptide molecules (or consist of multiple polypeptide subunits). The distinction is that peptides are short and polypeptides/proteins are long.
C12: Differentiate among the following levels of protein organization.
Primary Structure. Proteins are made up of polypeptide chains, which are amino acids joined together with peptide bonds. The unique sequence of amino acids that make up a protein or polypeptide chain is called the Primary Structure.
Secondary Structure: After synthesis, polypeptide chains are folded or pleated into different shapes, called their Secondary Structure. Two common examples of secondary structures are Alpha Helices and Beta Pleated Sheets. Secondary structure is held together by many Hydrogen bonds, overall giving the shape great stability.
Tertiary Structure: The final 3D structure of a protein is its Tertiary Structure, which pertains to the shaping of the secondary structure. This may involve coiling or pleating, often with straight chains of amino acids in between.
Quaternary Structure: The structure formed when two or more polypeptide chains join together, sometimes with an inorganic component, to form a protein.
C13: List the major functions of of proteins.
Protein is essential to the structure of red blood cells, for the proper functioning of antibodies resisting infection, for the regulation of enzymes and hormones, for growth, and for the repair of body tissue. Protein sources: Amino acids are the building blocks of protein and are found in a variety of foods.
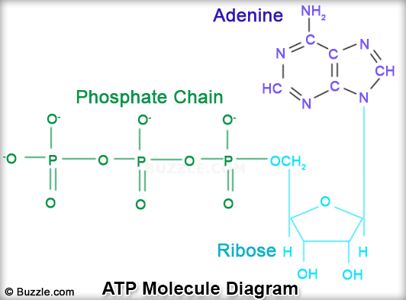
C14: Relate the general structure of the ATP molecule to its role as the "energy currency" of cells.
ATP (adenosine triphosphate) is a nucleotide that functions as an energy carrier in cells. In ATP, the base adenine is joined to the sugar ribose (together called adenosine), and there are three phosphate groups (triphosphate) instead of one. When one of the phosphate groups are removed, a large amount of energy is released.
Peptides are short polymers formed from the linking, in a defined order, of α-amino acids. The link between one amino acid residue and the next is known as an amide bond or a peptide bond.
A dipeptide is a molecule consisting of two amino acids joined by a single peptide bond.
Proteins are polypeptide molecules (or consist of multiple polypeptide subunits). The distinction is that peptides are short and polypeptides/proteins are long.
C12: Differentiate among the following levels of protein organization.
Primary Structure. Proteins are made up of polypeptide chains, which are amino acids joined together with peptide bonds. The unique sequence of amino acids that make up a protein or polypeptide chain is called the Primary Structure.
Secondary Structure: After synthesis, polypeptide chains are folded or pleated into different shapes, called their Secondary Structure. Two common examples of secondary structures are Alpha Helices and Beta Pleated Sheets. Secondary structure is held together by many Hydrogen bonds, overall giving the shape great stability.
Tertiary Structure: The final 3D structure of a protein is its Tertiary Structure, which pertains to the shaping of the secondary structure. This may involve coiling or pleating, often with straight chains of amino acids in between.
Quaternary Structure: The structure formed when two or more polypeptide chains join together, sometimes with an inorganic component, to form a protein.
C13: List the major functions of of proteins.
Protein is essential to the structure of red blood cells, for the proper functioning of antibodies resisting infection, for the regulation of enzymes and hormones, for growth, and for the repair of body tissue. Protein sources: Amino acids are the building blocks of protein and are found in a variety of foods.
C14: Relate the general structure of the ATP molecule to its role as the "energy currency" of cells.
ATP (adenosine triphosphate) is a nucleotide that functions as an energy carrier in cells. In ATP, the base adenine is joined to the sugar ribose (together called adenosine), and there are three phosphate groups (triphosphate) instead of one. When one of the phosphate groups are removed, a large amount of energy is released.